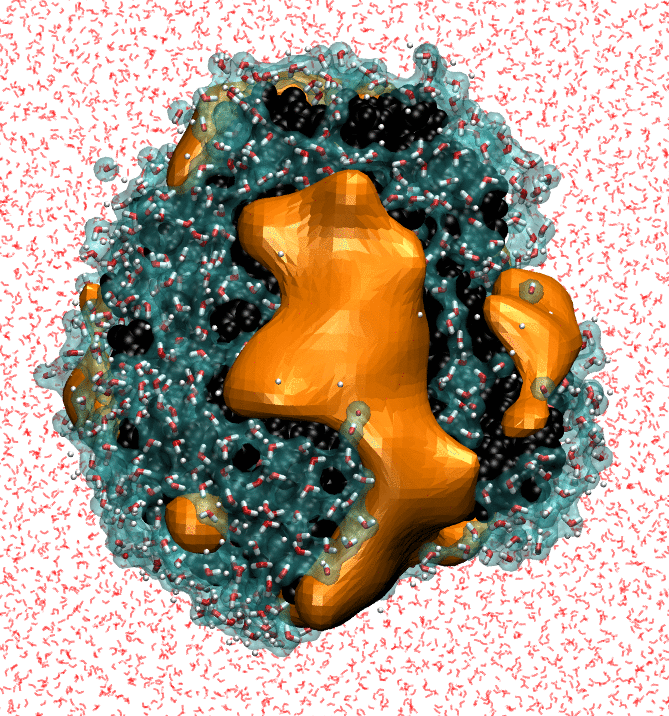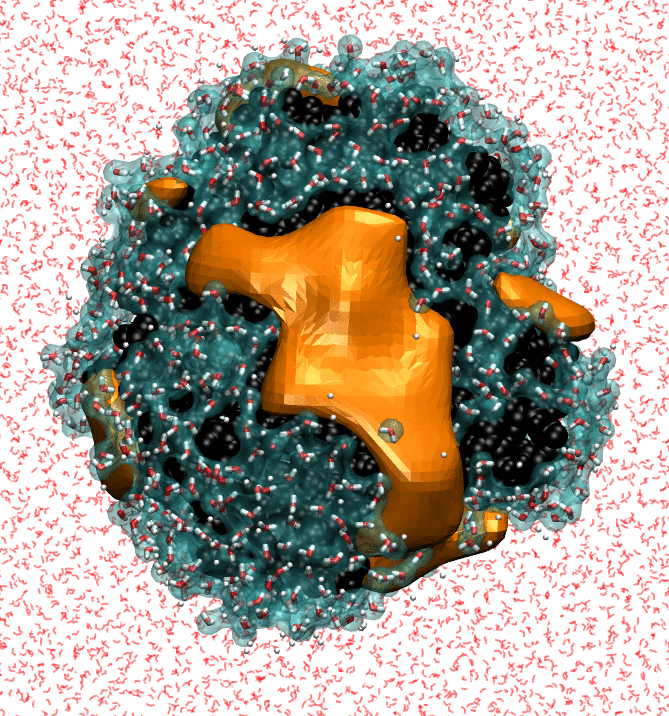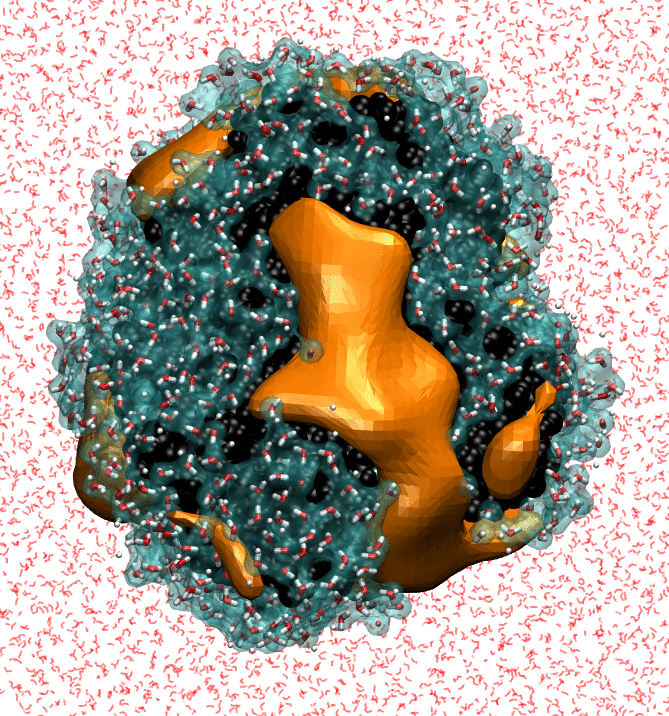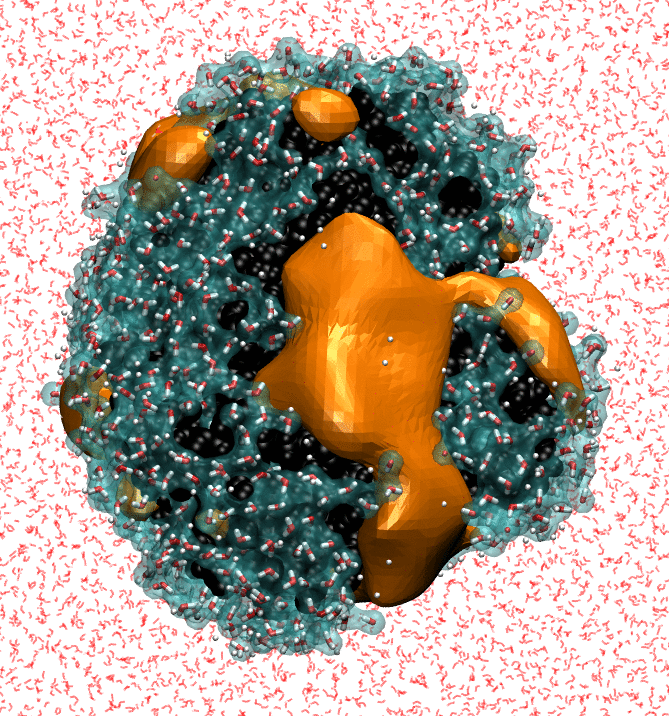
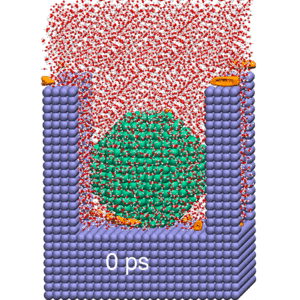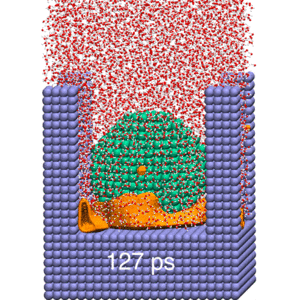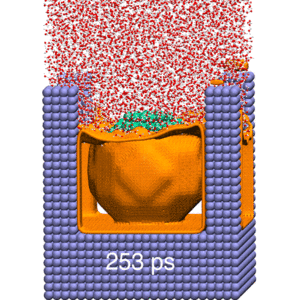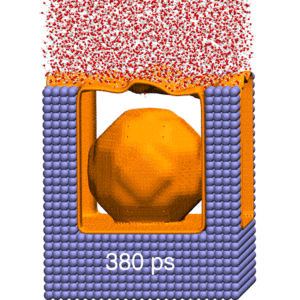
Our research strives to achieve a molecular-level understanding of solvation and transport in aqueous and polymeric systems, with applications ranging from the prediction of protein interactions to the design of advanced materials for water purification and renewable energy.
To study these biological, nanoscopic, and polymeric systems, our group combines principles of statistical mechanics and liquid state theory with state-of-the-art molecular modeling and atomistic simulation techniques. We interface closely with experimentalists, both to realize our predictions and to refine our theoretical models.
- Zac, Lilia, and Jun defended their dissertations. Congratulations!
- Lizhu presented at the Foundations of Molecular Modeling and Simulations Conference.
- Yusheng and Jeongmoon presented at the GRC on Water and Aqueous Solutions.
- Amish was promoted to full Professor.
- Jeongmoon received the Penn Prize for Excellence in Graduate Teaching.
- Akash, Amish, and Lizhu presented at the GRC on Chemistry and Physics of Liquids.
- PLUMED INDUS is publicly available, find it here: INDUS Code
Recent publications View all
BQ Kim, Z Vicars, M Füredi, LF Escobedo, RB Venkatesh, S Guldin, AJ Patel, and D Lee. "Amphiphilic nanopores that condense undersaturated water vapor and exude water droplets", Science Advances (2025).
Paper Media Coverage
CW Johnson, L Zhang, KE Culley, SM Oh, DL Gin, GM Geise, AJ Patel, KI Winey, and CO Osuji. "The Effects of Morphology and Hydration on Anion Transport in Self-Assembled Nanoporous Membranes", ACS Nano (2025).

Paper
NB Rego, AL Ferguson and AJ Patel. "Learning the relationship between nanoscale chemical patterning and hydrophobicity", Proceedings of the National Academy of Sciences, 119 (48) e2200018119 (2022).
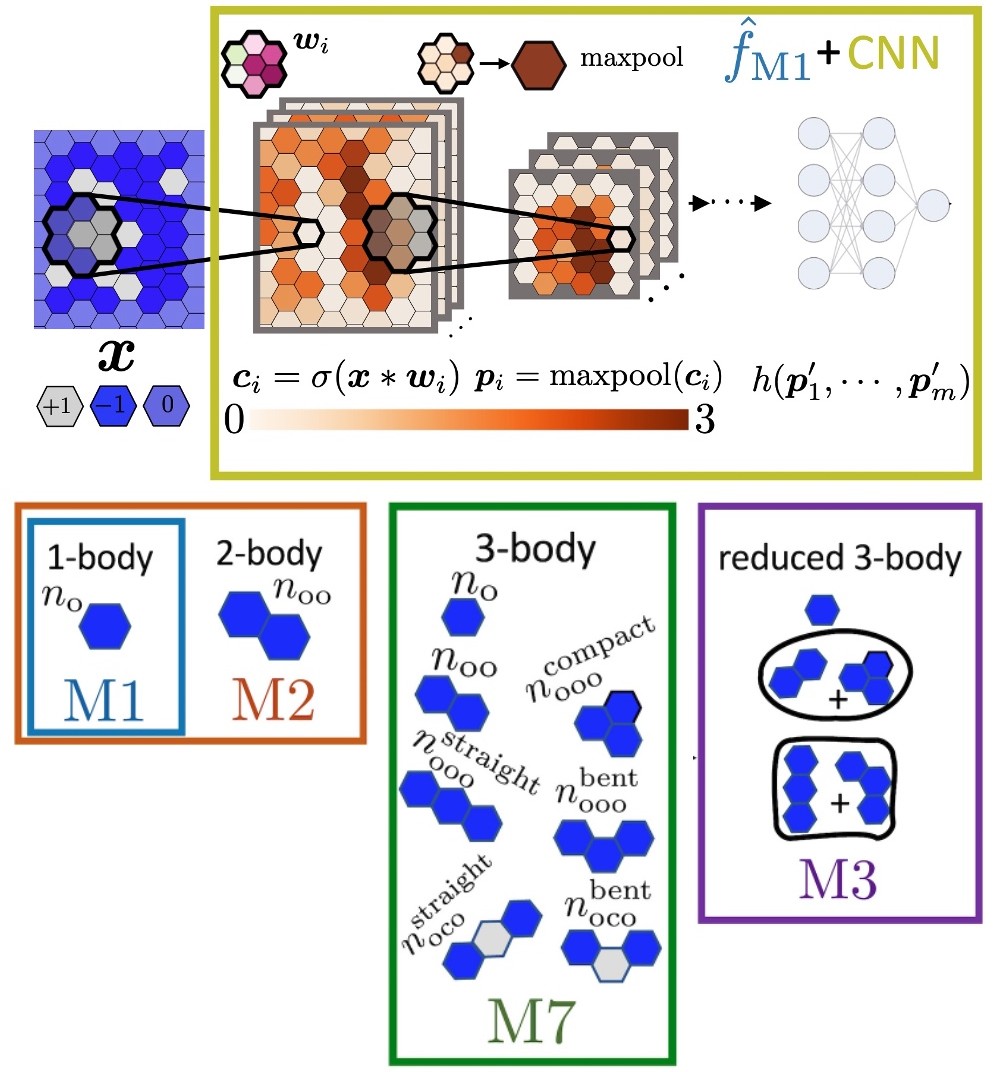
Paper
NB Rego and AJ Patel. "Understanding Hydrophobic Effects: Insights from Water Density Fluctuations", Annual Review of Condensed Matter Physics, 13 (1), 303–324 (2022).
Paper arXiv Preprint
AU Thosar and AJ Patel. "Hydration determines anion accumulation", Nature Chemistry, 14 (1), 8–10 (2022).
Paper
D Dhabal, Z Jiang, A Pallath, and AJ Patel. "Characterizing the Interplay between Polymer Solvation and Conformation", Journal of Physical Chemistry B, 125 (20), 5434–5442 (2021).
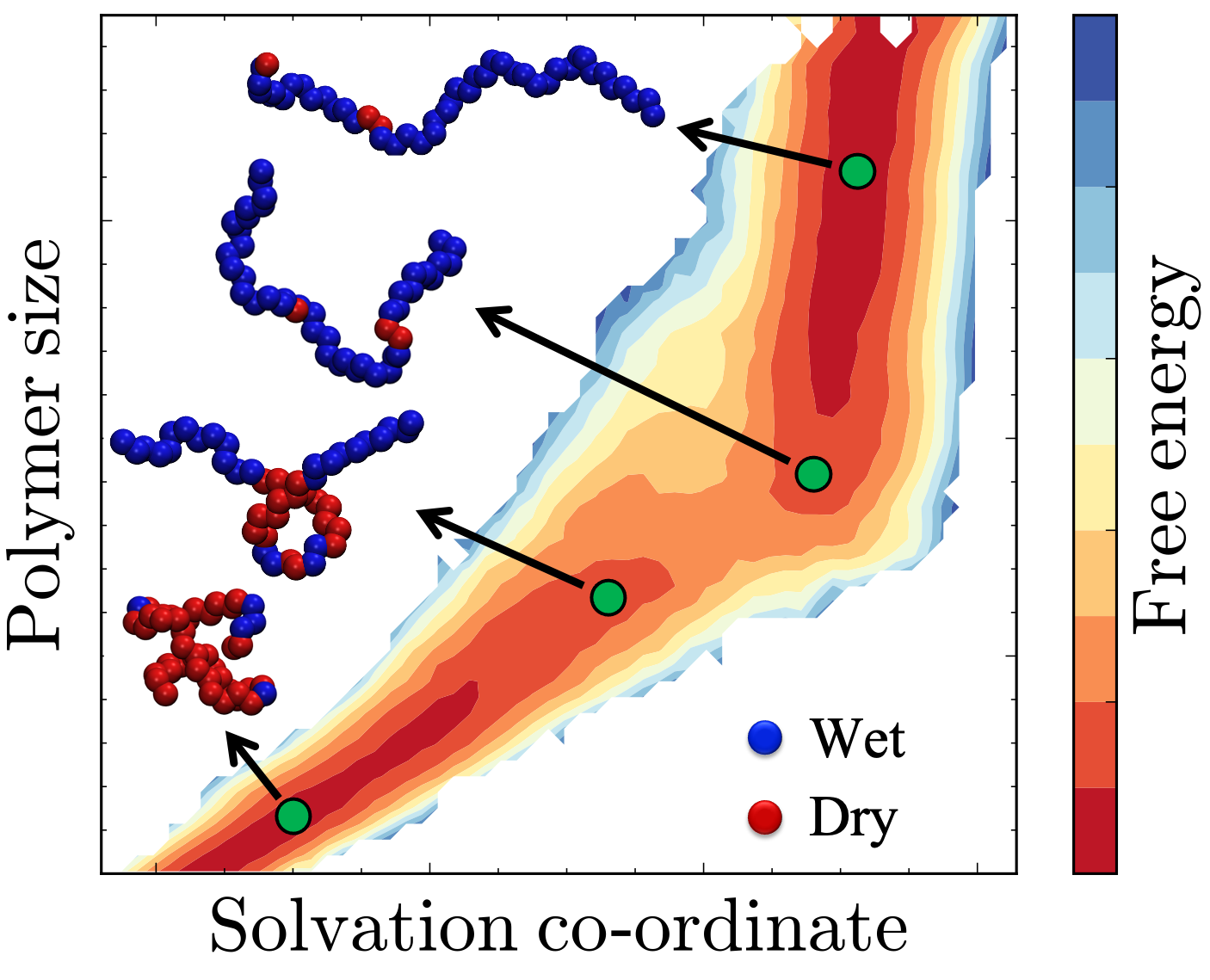
Paper arXiv Preprint
UR Gabinet, C Lee, R Poling-Skutvik, D Keane, NK Kim, R Dong, Z Vicars, Y Cai, AU Thosar, A Grun, SM Thompson, AJ Patel, CR Kagan, RJ Composto, and CO Osuji. "Nanocomposites of 2D-MoS2 Exfoliated in Thermotropic Liquid Crystals", ACS Materials Letters, 3(6), 704–712 (2021).
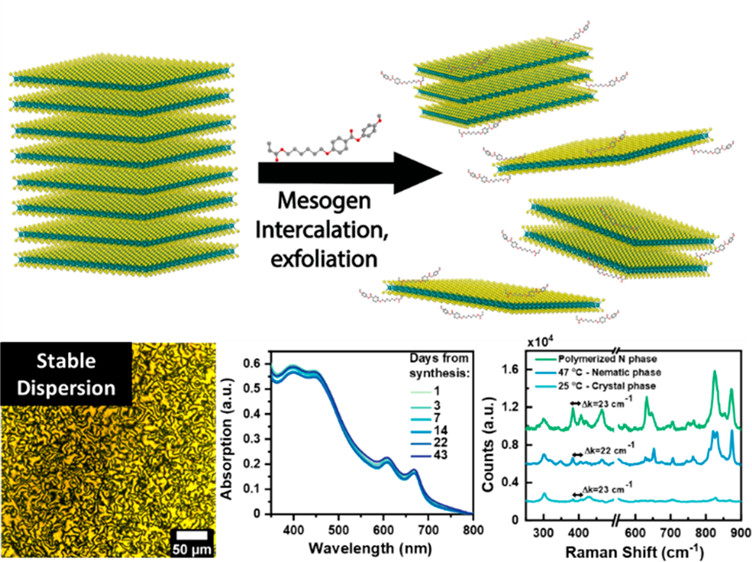
Paper
NB Rego, E Xi, and AJ Patel. "Identifying Hydrophobic Protein Patches to Inform Protein Interaction Interfaces", Proceedings of the National Academy of Sciences, 118 (6) e2018234118 (2021).

Paper