
Nicholas Rego
Research interests
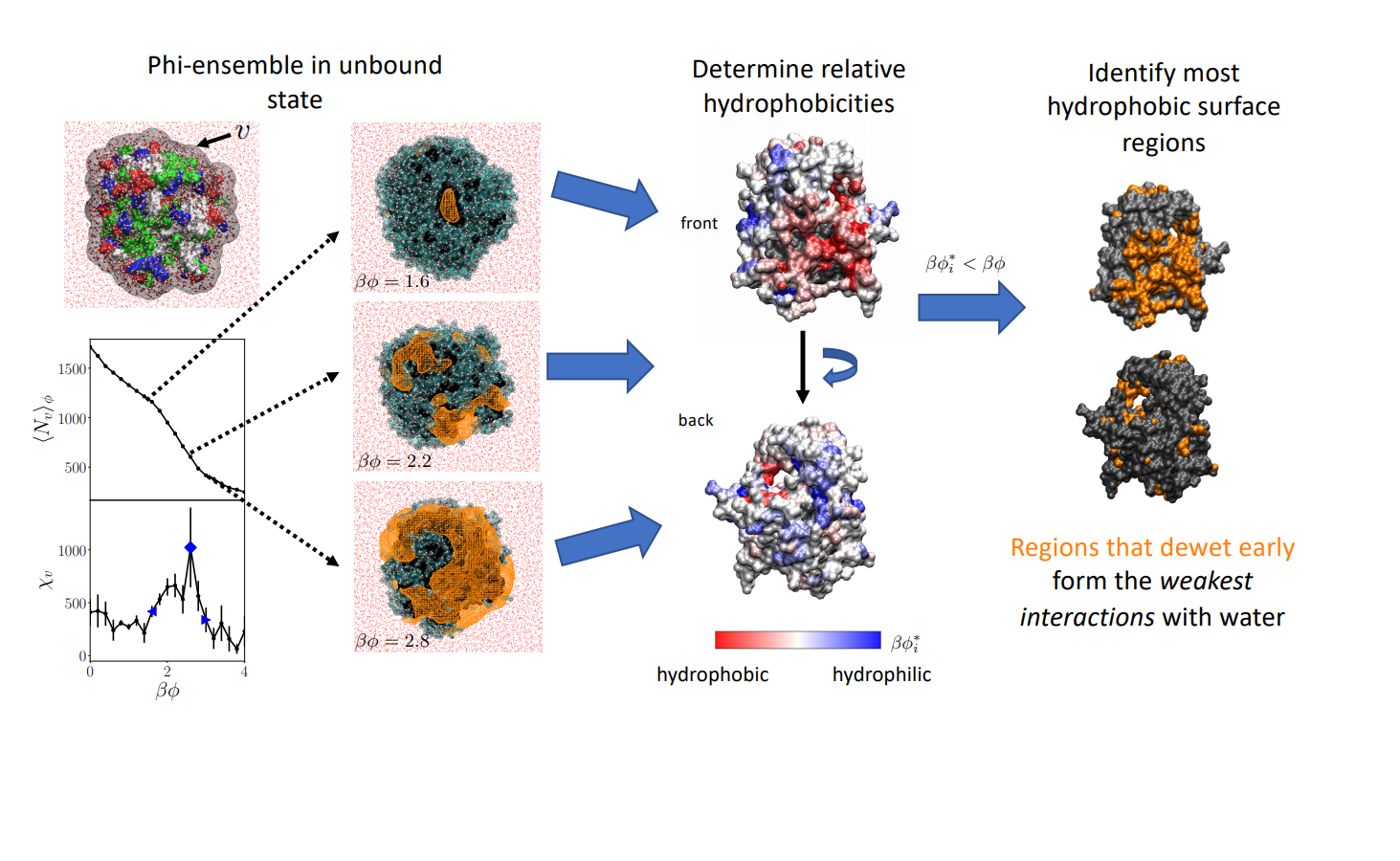
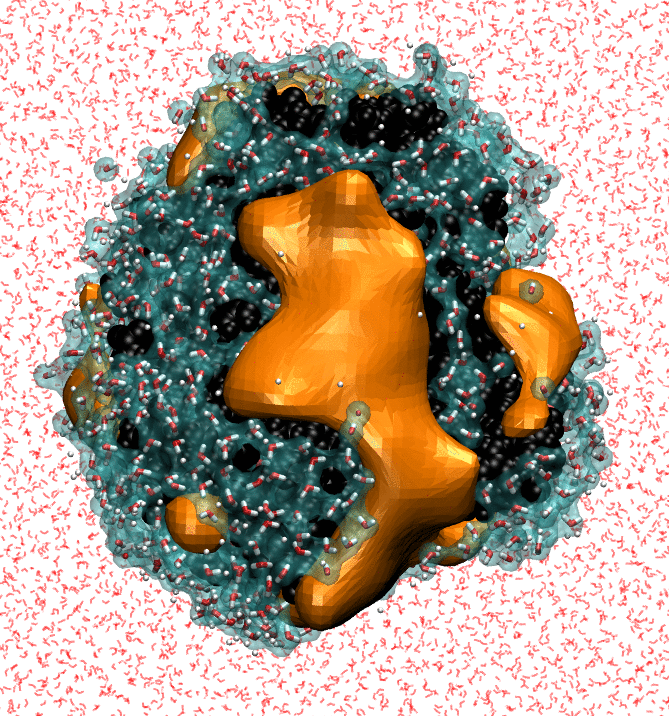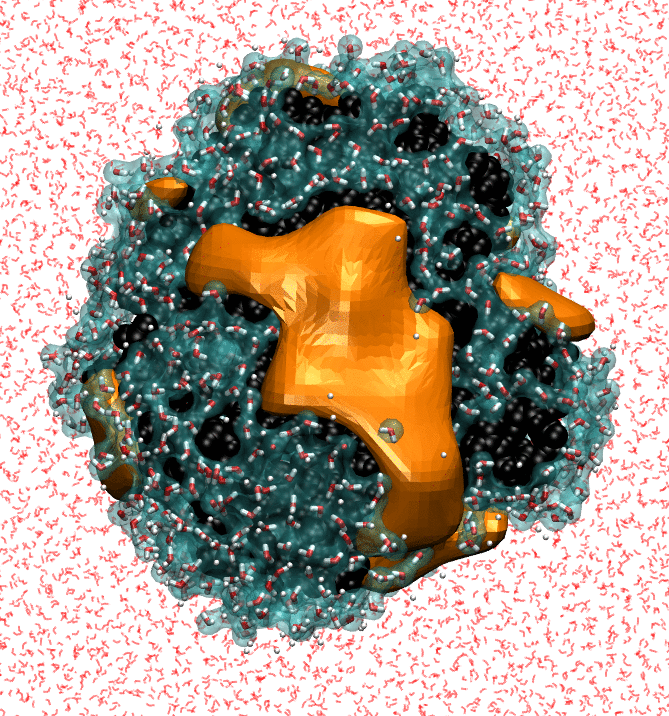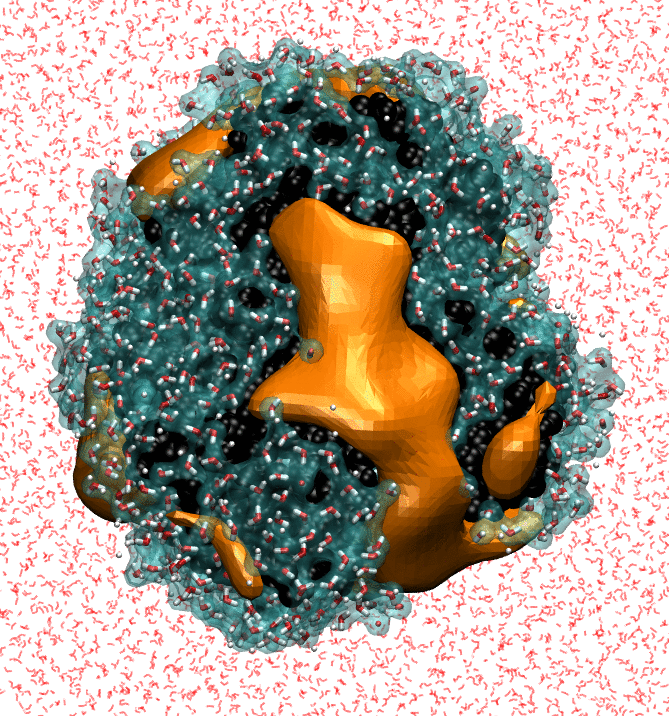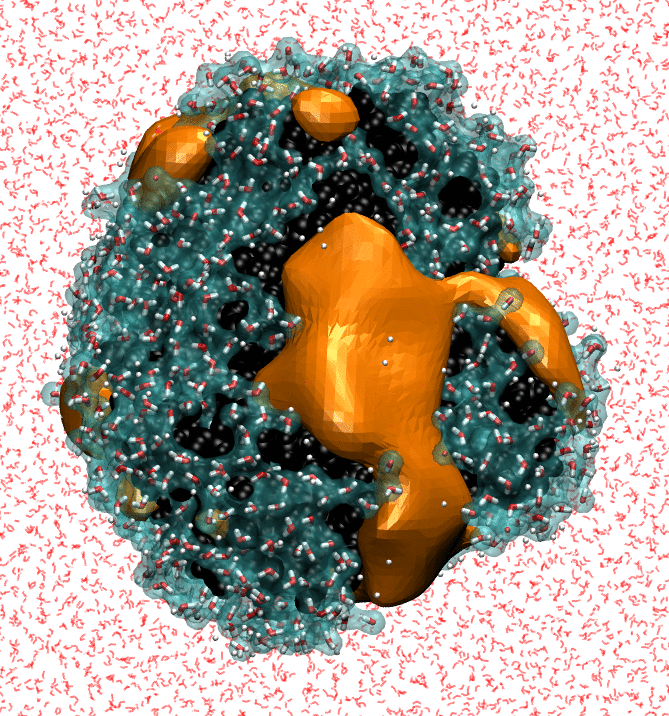
My research focuses on characterizing the strength of water-protein interactions. The surfaces of proteins are extremely complex, both chemically and topographically, and the ways in which proteins disrupt hydrating water structure determines their interaction sites, and, ultimately, their functions. Characterizing the strength of protein surface-water interactions is challenging; we do this by systematically perturbing protein hydration waters to identify surface patches that interact the most weakly with water. Finally, I'm trying to determine the underlying topographical and chemical determinants of hydrophobicity at the nanoscale by leveraging interpretable machine-learning techniques on simple model surfaces.
Publications
- NB Rego, AL Ferguson and AJ Patel. "Learning the relationship between nanoscale chemical patterning and hydrophobicity", Proceedings of the National Academy of Sciences (2022).
- NB Rego, AJ Patel. "Understanding Hydrophobic Effects: Insights from Water Density Fluctuations", Annual Review of Condensed Matter Physics (2022). arXiv
- NB Rego, E Xi, AJ Patel. "Identifying Hydrophobic Protein Patches to Inform Protein Interaction Interfaces", Proceedings of the National Academy of Sciences, 118 (6) e2018234118 (2021).
- Z Jiang, R Remsing, NB Rego, AJ Patel. "Characterizing Solvent Density Fluctuations in Dynamical Observation Volumes", Journal of Physical Chemistry B, 123, 7, 1650-1661 (2019). arXiv
- NB Rego, E Xi, AJ Patel. "Protein Hydration Waters are Susceptible to Unfavorable Perturbations", Journal of the American Chemical Society, 141, 2080-2086 (2019). arXiv
- E Xi, V Venkateshwaran, L Li, NB Rego, AJ Patel, and S Garde, Hydrophobicity of proteins and nanostructured solutes is governed by topographical and chemical context, Proceedings of the National Academy of Sciences, 114, 13345-13350 (2017).